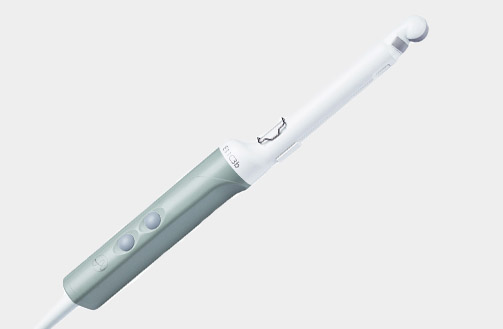
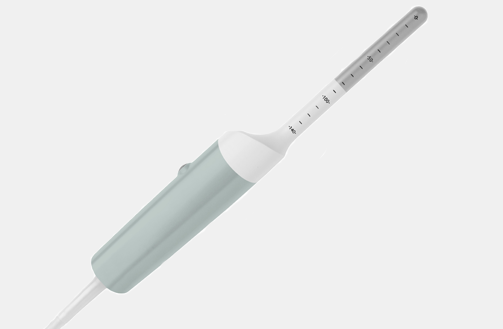
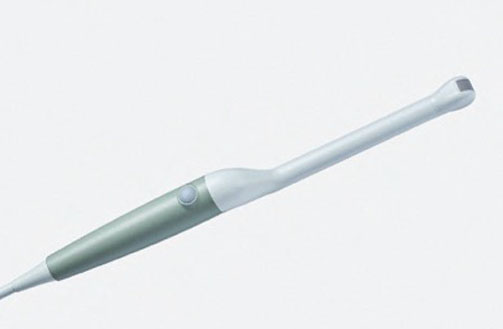
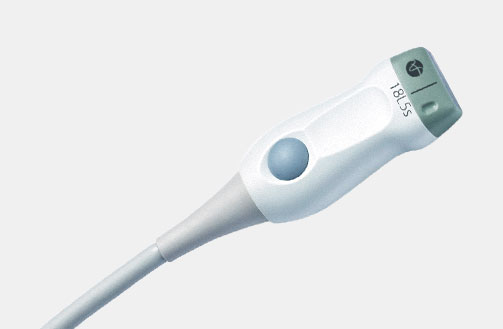
Cleaning Information
Correct reprocessing is fundamental in guarding against infection. To make liquid-based reprocessing more convenient, most BK Medical transducers come with a special watertight plug lid to allow the entire transducer (including the cable and plug) to be fully submerged.
BK Medical equipment is designed to be durable but as devices are complex, care is needed when handling them.
BK Medical continuously validates and evaluates different reprocessing agents and methods.
To download the care and cleaning guide with reprocessing posters, select your language
Care and Cleaning Methods for Transducers
Follow local regulations for minimum reprocessing. Check table 4 on page 29 of the Care and Cleaning Guide.
Follow product manufacturer’s instructions and do not exceed transducer specified limits. See Care and Cleaning Guide for more information.
Click the transducer to read the information.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
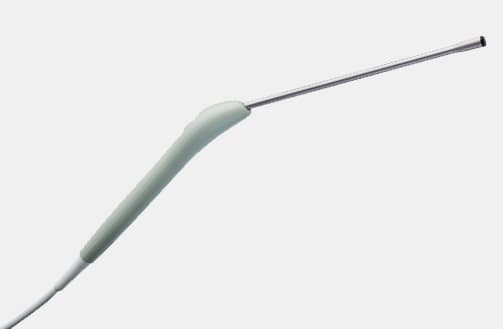
N20P6 (9007) Minimally Invasive Transducer
d
Footnote:
d) Transducer has not been licensed by Health Canada.
 Print Cleaning Methods
Print Cleaning Methods
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
5P1e (9087) Phased Array Transducer
b
Footnote:
b) Transducer connector is not immersible.
 Print Cleaning Methods
Print Cleaning Methods
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
14L3e (9086) Linear Transducer
b
Footnote:
b) Transducer connector is not immersible.
 Print Cleaning Methods
Print Cleaning Methods
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
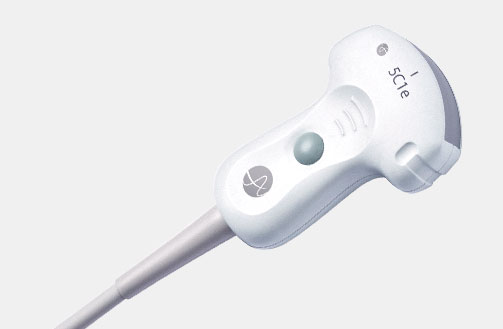
5C1e (9085) Curved Array Transducer
b
Footnote:
b) Transducer connector is not immersible.
 Print Cleaning Methods
Print Cleaning Methods
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Efficacy validated by BK
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
Material Compatible
a
Footnote:
a) “Material compatible” indicates that BK Medical has evaluated the device’s material compatibility with the reprocessing method when reprocessed according to the product/system IFU. Efficacy is not covered by this statement.
We're sorry, we couldn't find any results matching that query.